Vous êtes ici :
- Unité de recherche
- BioCIS
- Accueil
- Publications
- Articles
- 2023-2022
2023-2021
- 2023
-
1. Development and qualification of clinical grade decellularized and cryopreserved human esophagi, Godefroy, W.; Faivre, L.; Sansac, C.; Thierry, B.; Allain, J.-M.; Bruneval, P.; Agniel, R.; Kellouche, S.; Monasson, O.; Peroni, E.; Jarraya, M.; Setterblad, N.; Braik, M.; Even, B.; Cheverry, S.; Domet, T.; Albanese, P.; Larghero, J.; Cattan, P.; Arakelian, L.; Sci Rep, 2023, 18283.
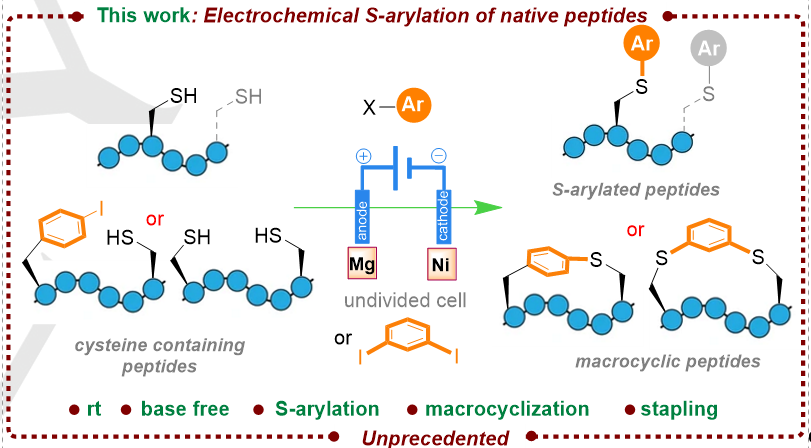
2. Electrochemical Nickel-Catalyzed Selective Inter- and Intramolecular Arylations of Cysteine-Containing Peptides, Shen, L.; Monasson, O.; Peroni, E.; Lebideau, F.; Messaoudi, S.; Angew. Chem. Int. Ed., 2023, e202315748
acie
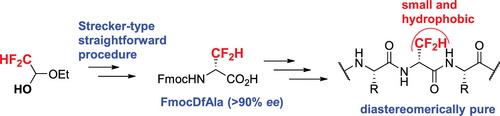
3.Difluoroalanine: Synthesis, Incorporation in Peptides, and Hydrophobic Contribution Assessment, Boutahri, Y.; Ben Haj Salah, K.; Tisserand, N.; Lensen, N.; Crousse, B.; Brigaud, T.; Org.Lett., 2023, 25, 37, 6937–6941ol 2023
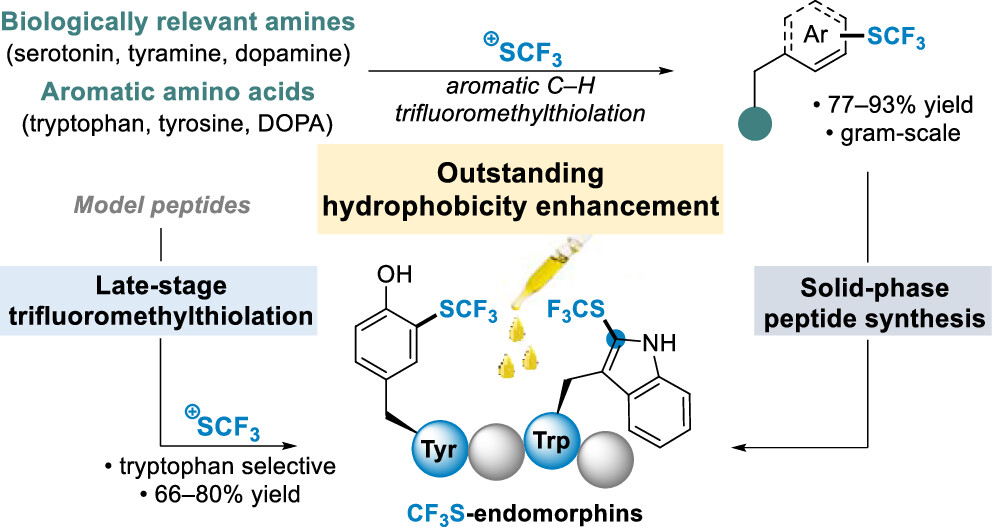
4. Trifluoromethylthiolation of Tryptophan and Tyrosine Derivatives: A Tool for Enhancing the Local Hydrophobicity of Peptides, Gregorc, J.; Lensen, N.; Chaume, G.; Iskra, J.; Brigaud, T.; J. Org.Chem., 2023, 88, 18, 13169-13177
joc 2023
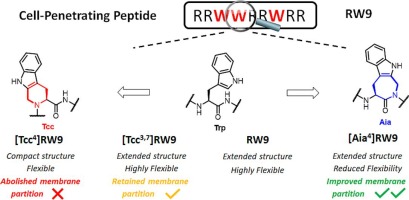
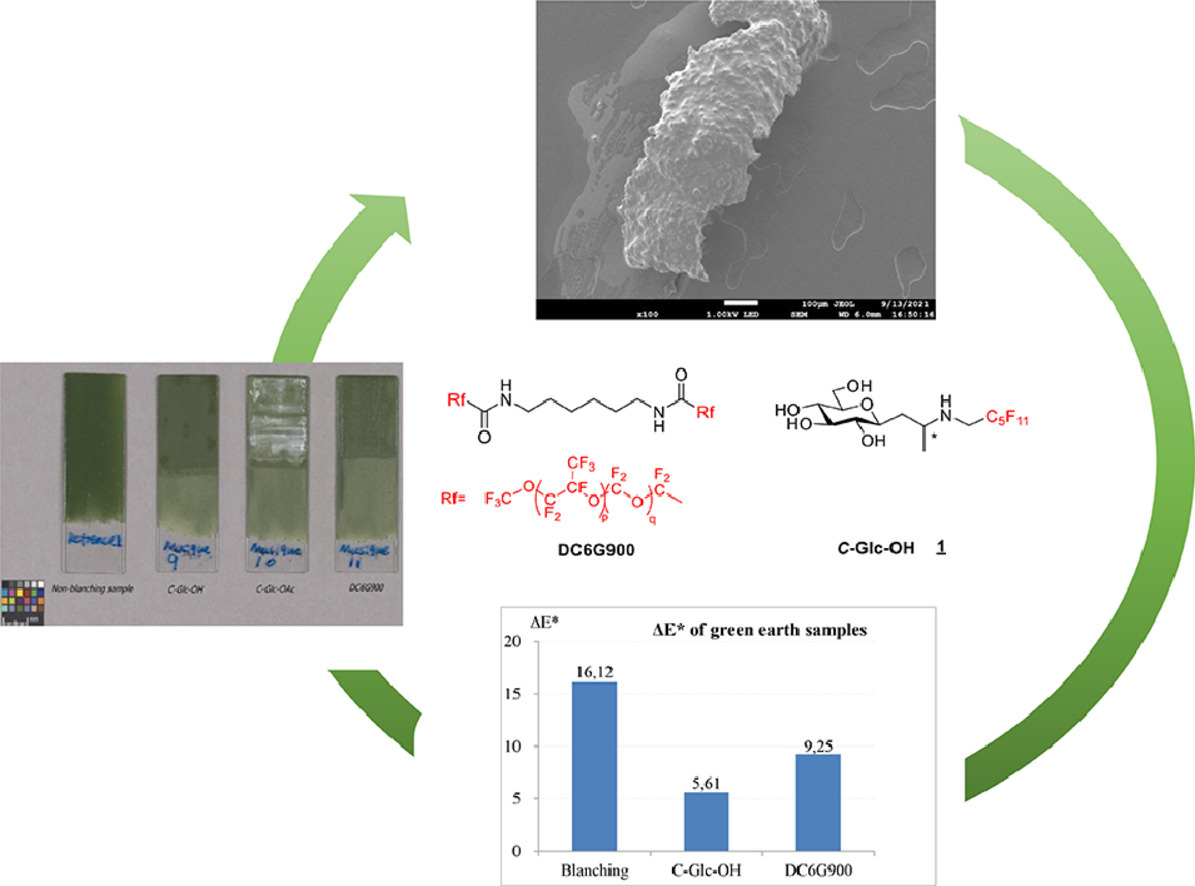
5. Introduction of constrained Trp analogs in RW9 modulates structure and partition in membrane models, Lozada, C.; Gonzalez, S.; Agniel, R.; Hindie, M.; Manciocchi, L.; Mazzanti, L.; Ha-Duong, T.; Santoro, F.; Carotenuto, A.; Ballet, S.; Lubin-Germain, N.; Biorganic Chemistry, 2023, 139, 106731Lozada 6. Synthesis of novel partially perfluorinated C-glycosides and their application in blanching easel painting restoration, Zhang, Y.; Eveno, M.; Gallier, F.; Camaiti, M. Lattuati-Derieux, A.; Salvini, A.; Lubin-Germain, N.; Journal of Cultural Heritage, 2023, 62, 493-500
cult her
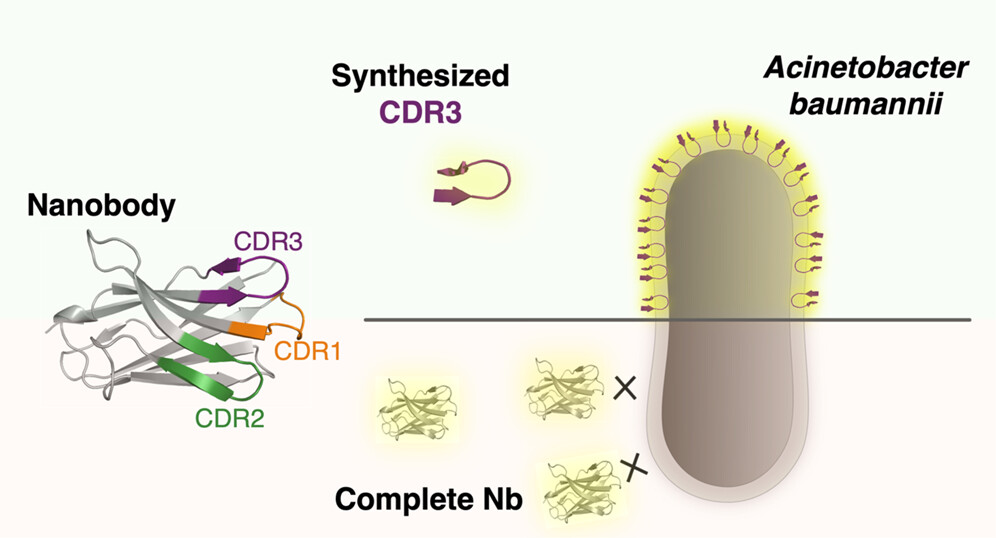
7. Bypassing the Need for Cell Permeabilization: Nanobody CDR3 Peptide Improves Binding on Living Bacteria, Breine, A.; Van holsbeeck, K.; Martin, C.; Gonzalez, S.; Mannes, M.; Pardon, E.; Steyaert, J.; Remaut, H.; Ballet, S.; Van der Henst, C.; BioconjugateChem. 2023, 34,1234−1243gonz
8. Time-of-flight SIMS investigation of peptides containing cell penetrating sequences, Auditore, A.; Tuccitto, N.; Grasso, G.; Monasson, O.; Peroni, E.; Licciardello, A.; Biointerphases. 2023, 18, 031006
9. Inviting C5-Trifluoromethylated Pseudoprolines into Collagen Mimetic Peptides, Terrien, A.; Rahgoshay, K.; Renaglia, E.; Lensen, N.; Jacquot, Y.; Marquant, R.; Brigaud, T.; Loison, C.; Chaume, G.; Miclet, E.; Biomacromolecules, 2023, 24, 4, 1555–1562graph biomacro 2023 - 2022
-
1. Studies on the synthesis of 1'-CN-triazolyl-C-ribosides, Bozinovic, N.; Marques de Aguiar, V.; Ferry, A.; Gallier, F.; Lubin-Germain, N.; Uziel, J.; Soter de Mariz e Miranda, L.; Org.Biomol.Chem, 2022, 7261-7269.
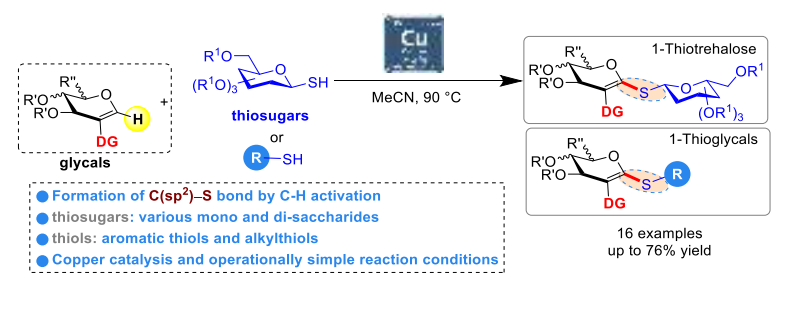
2. Directed dehydrogenative copper-catalyzed C-H thiolation in pseudo-anomeric position of glycals using thiol and thiosugar partners, Li, L.; Mahri, L.; de Robichon, M.; Fatthalla, M.; Ferry, A.; Messaoudi, S.; Adv. Synth. Catal. 2022, ASAP articleASC ferry
3. Low Fluorinated Oligoamides for Use as Wood Protective, Zhang, Y.; Vespignani, L.; Balzano, M. G.; Bellandi, L.; Camaiti, M.; Lubin-Germain, N.; Salvini, A. Coatings 2022, 12 (7), 927.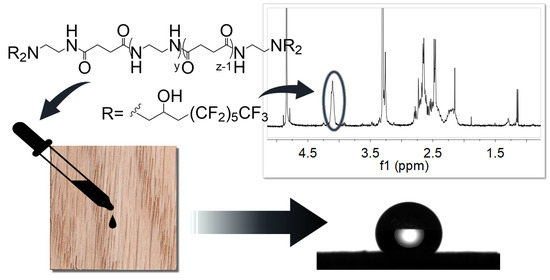
4. Carbon dots surface chemistry drives fluorescent properties: New tools to distinguish isobaric peptides, Distefano, A.: Cali, F.; Gaeta, M.; Tuccitto, N.; Auditore, A.; Licciardello, A.; D'Urso, A.; Lee, K.-J.; Monasson, O.; Peroni, E.; Grasso, G.; J. Colloid Interface Sci., 2022, 625, 405
5. Anti-HCV and Zika activities of ribavirin C-nucleosides analogues, Gonzalez, S.; Brzuska, G.;Ouarti, A.; Gallier, F.; Solarte, C.; Ferry, A.; Uziel, J.; Krol, E.; Lubin-Germain, N.; Bioorg. Med. Chem, 2022, 68, 116858
6. Highly stereoselective multigram scale synthesis of (3S,4S)-Statine and (3S,4S)-N-Benzylstatine, Ben Haj Salah, K; Terzani, F.; Pietri, M.; Zanato, C.; Chelain, E.; Pytkowicz, J.; Results in Chemistry, 2022, 4, 100333.
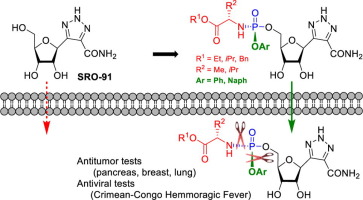
7. Synthesis, antiviral and antitumor activities investigations of a series of Ribavirin C-nucleoside analogue prodrugs, Sabat, N.; Ouarti, A.; Migianu-Griffoni, E.; Lecouvey, M.; Ferraris, O.; Gallier, F.; Peyrefitte, C.; Lubin-Germain, N.; Uziel, J.; Bioorganic Chemistry, 2022, 122, 105723graph abs bioorgchem 2022
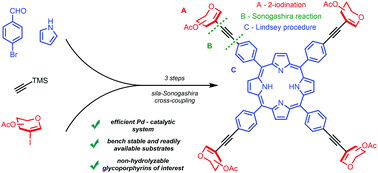
8. Sonogashira cross-coupling as a key step in the synthesis of new glycoporphyrins, Godlewski, B.; Baran, D.; de Robichon, M.; Ferry, A.; Ostrowski, S.; Malinowski, M.; Org. Chem. Front., 2022, 2396-2404
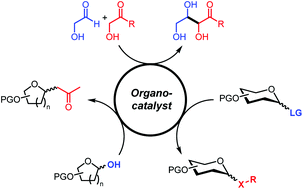
9. Organocatalysis applied to carbohydrates: from roots to current developments, Florian Gallier and Leandro Soter de Mariz e Miranda, Org. Biomol. Chem., 2022,20, 919-93310. Hirao cross-coupling reaction as efficient tool to build non-natural C2-phosphonylated sugars, Monasson, O.; Malinowski, M.; Lubin-Germain, N.; Ferry, A.; Synthesis, 2022, 3414-3420

11. Introducing the chiral constrained α-trifluoromethylalanine in aib foldamers to control, quantify and assign the helical screw-sense, Bodero, L., Guitot, K., Lensen, N., Lequin, O., Brigaud, T., Ongeri, S., Chaume, G. Chemistry - A European Journal, 2022, 28 (8), e202103887
12. (R)-α-Trifluoromethylalanine as a 19F NMR Probe for the Monitoring of Protease Digestion of Peptides. Devillers, E.; Chelain, E.; Dalvit, C.; Brigaud, T.; Pytkowicz, J., ChemBioChem 2022,23, e202100470

- 2021
-
1. Copper and Nickel Nanoparticles Prepared by Thermal Treatment of Their Respective Cations Confined in Nanopores through High-Pressure Synthesis. Brodie-Linder,N.; Deschamps, J.; Bombled, M.; Pasternak, N.; Audonnet, F.; Beaunier, P.; Alba-Simionesco, C. Appl. Nano 2021, 278-288
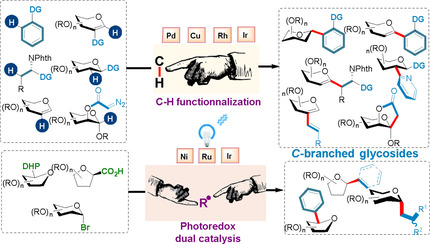
2. Emerging Organometallic Methods for the Synthesis of C-Branched (Hetero)aryl, Alkenyl, and Alkyl Glycosides: C-H Functionalization and Dual Photoredox Approaches, Ghouilem, J.; de Robichon, M.; Le Bideau, F.; Ferry, A.; Messaoudi, S; Chemistry - A European Journal, 2021, 27(2), pp. 491–511
graph abs chemistry 2021
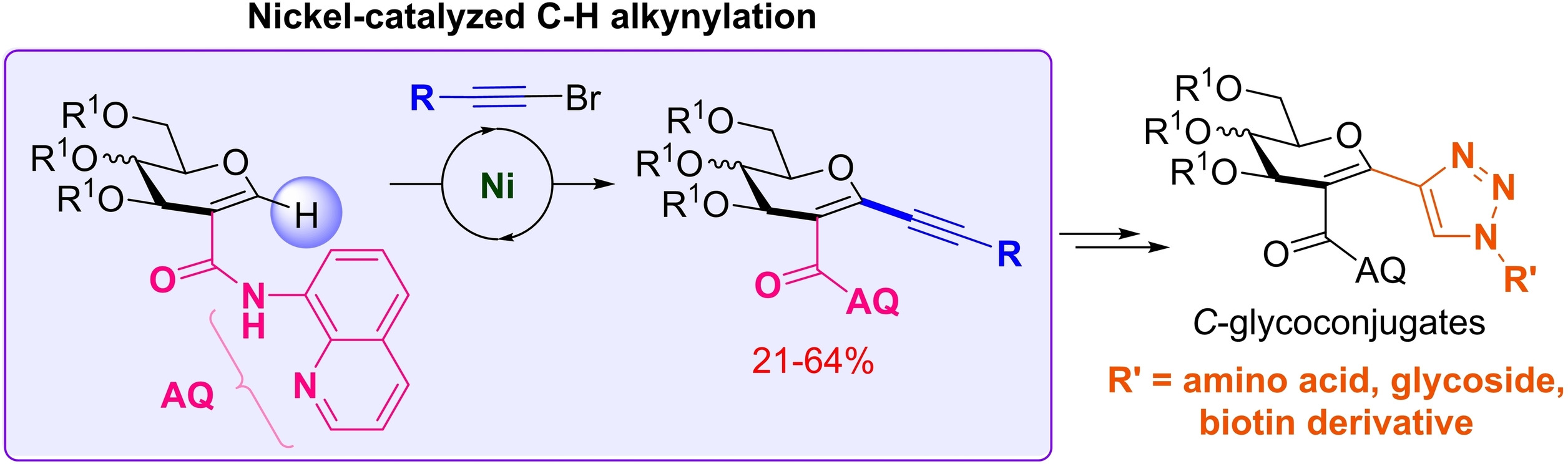
3. Directed Nickel-Catalyzed Pseudo-Anomeric C−H Alkynylation of Glycals as an Approach towards C-Glycoconjugate Synthesis. de Robichon, M.; Branquet, D.; Uziel, J.; Lubin-Germain, N.; Ferry, A. Adv. Synth. Catal. 2021,363 (22), pp. 5138-5148
asc 2021
4. Identification and characteristics of fusion peptides derived from enveloped viruses C. Lozada, T. M. A. Barlow, S. Gonzalez, N. Lubin-Germain, S. Ballet Front. Chem. 2021, 9:689006
5. Synthesis of enantiopure α-Tfm-proline and α-Tfm-pipecolic acid from oxazolo-pyrrolidines and -piperidines. C. A. Sanchez, C. Gadais, S. Diarra, A. Bordessa, N. Lensen, E. Chelain and T. Brigaud, Org. Biomol. Chem., 2021, 6771-6775
6. Synthesis of the Fungal Metabolite YWA1 and Related Constructs as Tools to Study MelLec-Mediated Immune Response to Aspergillus Infections. Piras, M.; Patruno, I.; Nikolakopoulou, C.; Willment, J. A.; Sloan, N. L.; Zanato, C.; Brown, G. D.; Zanda, M. J. Org. Chem. 2021, 6044–6055

7. Glycosamine Derivatives through Metal-Catalyzed C−N Bond Formation on Protected and Unprotected 2-Iodoglycals. Malinowski, M.; Banoun, C.; de Robichon, M.; Lubin-Germain, N.; Ferry, A. Eur. J. Org. Chem. 2021, 1521–1524.

8. Enantiopure 5-CF3–Proline: Synthesis, Incorporation in Peptides, and Tuning of the Peptide Bond Geometry. C. A. Sanchez, C. Gadais, C.;Chaume, G.; Girard, S.; Chelain, E.; Brigaud, T. Org. Lett. 2021, 382-387