You are here :
- Unité de recherche
- BioCIS
- Home
- Publications
- Articles
- 2020-2016
2020-2016
- 2020
-
1. Mild Palladium‐Catalyzed Cyanation of Unprotected 2‐Iodoglycals in Aqueous Media as Versatile Tool to Access Diverse C2‐Glycoanalogues. Malinowski, M.; Tran, T. V.; de Robichon, M.; Lubin-Germain, N.; Ferry, A. Adv. Synth. Catal. 2020, 362, 1184-1189.

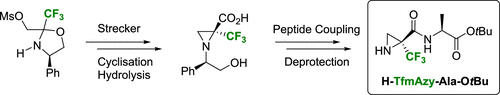
2. Enantiopure α-Trifluoromethylated Aziridine-2-carboxylic Acid (α-TfmAzy): Synthesis and Peptide Coupling. Ouerfelli, O.; Simon, J.; Chelain, E.; Pytkowicz, J.; Besbes, R.; Brigaud, T. Org. Lett. 2020, 22, 2946-2949.
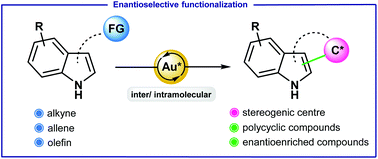
3. Gold-catalyzed enantioselective functionalization of indoles. Milcendeau, P.; Sabat, N.; Ferry, A. ; Guinchard, X. Org. Biomol. Chem. 2020, 18, 6006-6017.
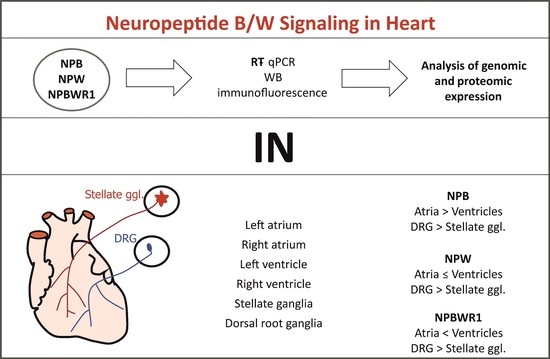
4. Identification of NPB, NPW and Their Receptor in the Rat Heart. Pandey, S.; Tuma, Z.; Peroni, E.; Monasson, O.; Papini, A. M.; Dvorakova, M. C. Int. J. Mol. Sci. 2020, 21, 7827
5. A Multiple N-Glucosylated Peptide Epitope Efficiently Detecting Antibodies in Multiple Sclerosis. Nuti, F., Fernandez, FR., Sabatino, G., Peroni, E., Mulinacci, B., Paolini, I., Di Pisa, M., Tiberi, C., Lolli, F., Petruzzo, M., Lanzillo, R., Brescia Morra, V., Rovero, P., Papini A. M. Brain Sci. 2020, 10, 453.
6. Hyperglucosylated adhesin-derived peptides as antigenic probes in multiple sclerosis: Structure optimization and immunological evaluation. Mazzoleni, A.,Real-Fernandez, F., Larregola, M., Nuti, F., Lequin, O., Papini, AM., Mallet, JM., Rovero, P. J. Pept. Sci. 2020, 26, e3281.
7. Direct Synthesis of Mesoporous Organosilica and Proof-of-Concept Applications in Lysozyme Adsorption and Supported Catalysis. Osta, O., Bombled, M., Partouche, D., Gallier, F., Lubin-Germain, N., Brodie-Linder, N., Alba-Simionesco, C. ACS Omega 2020, 5, 18842-18848.

8. Synthesis and antitumor activities investigation of a C-nucleoside analogue of ribavirin. Sabat, N., Migianu-Griffoni, E., Tudela, T., Lecouvey, M., Kellouche, S., Carreiras, F., Gallier, F., Uziel, J., Lubin-Germain, N. Eur. J. Med. Chem. 2020, 188, 112009

9.« Les pigments verts en Égypte au début de la Troisième Période intermédiaire : de l’objet à l’artisan ». Lucile Brunel-Duverger, Yvan Coquinot, Nancy Brodie-Linder et Sandrine Pagès-Camagna Techne 50 2020-2 - 2019
- 1. Studies of membranotropic and fusogenic activity of two putative HCV fusion peptides. Gonzalez, S.; Gallier, F.; Kellouche, S.; Carreiras, F.; Novellino, E.; Carotenuto, A.; Chassaing, G.; Rovero, P.; Uziel, J.; Lubin-Germain, N. Biochim Biophys Acta Biomembr. 2019, 1861, 50–61.

2. A photochromic azobenzene peptidomimetic of a β-turn model peptide structure as a conformational switch. Papini, A. M.; Nuti, F.; Gellini, C.; Larregola, M.; Squillantini, L.; Chelli, R.; Salvi, P. R.; Lequin, O.; Pietraperzia, G. Front. Chem. - Supramolecular Chemistry, 2019, 7:180.
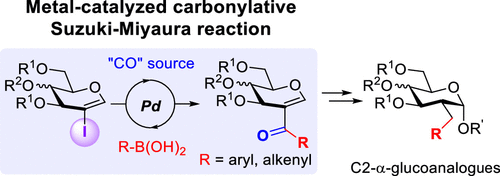
3. “CO” as a Carbon Bridge to Build Complex C2-Branched Glycosides Using a Palladium-Catalyzed Carbonylative Suzuki–Miyaura Reaction from 2-Iodoglycals. de Robichon, M.; Bordessa, A.; Lubin-Germain, N.; Ferry, A. J. Org. Chem. 2019, 84, 3328-3339.
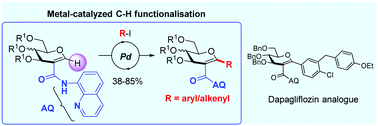
4. Access to C-aryl/alkenylglycosides by directed Pd-catalyzed C–H functionalisation of the anomeric position in glycal-type substrates. de Robichon, M.; Bordessa, A.; Malinowski, M.; Uziel, J.; Lubin-Germain, N.; Ferry, A. Chem. Commun. 2019, 55, 11806-11808.
5. CF2H as hydrogen bond donor group for the fine tuning of peptide bond geometry with difluoromethylated pseudoprolines. Malquin, N.; Rahgoshay, K.; Lensen, N.; Chaume, G.; Miclet, E.; Brigaud, T. Chem. Commun. 2019, 55, 12487-12490.
6. Trifluoromethylated Proline Surrogates as Part of "Pro-Pro" Turn-Inducing Templates. Gadais, C.; Van holsbeeck, K.; Moors, S. L. C.; Buyst, D.; Feher, K.; Van Hecke, K.; Tourwe, D.; Brigaud, T.; Martin, C.; De Proft, F.; Pytkowicz, J.; Martins, J. C.; Chaume, G.; Ballet, S. ChemBioChem 2019, 20, 2513-2518.

7. Photochemical Origin of the Darkening of Copper Acetate and Resinate Pigments in Historical Paintings M. Alter, L. Binet, N.Touati, N. Lubin-Germain, A.S.Le Hô, F. Mirambet, D.Gourier Inorg. Chem. 2019, 58, 19, 13115–13128.

8. Evaluation of the potential of a new ribavirin analog impairing the dissemination of ovarian cancer cells A. Wambecke, C. Laurent-Issartel, J. Leroy-Dudal, F. Giffard, F. Cosson, N.Lubin-Germain, J. Uziel, S. Kellouche, F. Carreiras Plos One 2019, 14 (12), e02225860.
9. The contribution of non-invasive and non-destructive techniques to the understanding of the 21st Dynasty Egyptian Yellow Coffins complex stratigraphy: Case of study of E 20043 from the Louvre Museum. Brunel-Duverger L., Laval E., Lemasson Q., Brodie-Linder N., Pages-Camagna, S. The European Physical Journal Plus 2019, 134, 257. - 2018
- 1. Orthogonal 19F-labeling for solid-state NMR reveals the conformation and orientation of short peptaibols in membranes. Grage, S. L.; Kara, S.; Bordessa, A.; Doan, V.; Rizzolo, F.; Putzu, M.; Kubař, T.; Papini, A. M.; Chaume, G.; Brigaud, T.; Afonin, S.; Ulrich, A. S. Chem. Eur. J. 2018, 24, 4328-4335

2. Probing the outstanding local hydrophobicity increase of peptide sequences induced by trifluoromethylated amino acids incorporation. Gadais, C.; Devillers, E.; Gasparik, V.; Chelain, E.; Pytkowicz, J.; Brigaud, T. ChemBioChem 2018, 19, 1026–1030.

3.Trifluoromethylated proline analogues as efficient tools to enhance the hydrophobicity and to promote passive diffusion transport of the L-prolyl-L-leucyl glycinamide (PLG) tripeptide. Oliver, M.; Gadais, C.; Garcia-Pindado, J.; Teixido, M.; Lensen, N.; Chaume, G.; Brigaud, T. RSC Adv. 2018, 8, 14597-14602.
4. Lipase-Catalyzed Amidation of Carboxylic Acid and Amines. Manova; D.; Gallier, F.; Tak-Tak, L.; Yotava, L.; Lubin-Germain, N. Tetrahedron Lett 2018, 59, 2086-2090.

5. Synthesis of C-pyrimidyl nucleosides starting from alkynyl ribofuranosides. Legrave, G.; Ait Youcef, R.; Afonso, D.; Ferry, A.; Uziel, J.; Lubin-Germain, N. Carbohydr. Res. 2018, 462, 50-55.

6. Antibodies to post-translationally modified mitochondrial peptide PDC-E2(167-184) in type 1 diabetes. Nuti, F.; Gallo, A.; Real-Fernandez, F.; Crulli, M.; Rentier, C.; Piarulli, F.; Peroni, E.; Rossi, G.; Traldi, P.; Rovero, P.; Lapolla, A.; Papini, A.M. Arch. Biochem. Biophys. 2018, 659, 66-74.
7. Stereoselective synthesis of 4-hydroxymethyl-1,3-oxazolidin-2-one derivatives from novel 2-hydroxymethylaziridines. Ouerfelli, O.; Ali Tabarki, M.; Pytkowicz, J.; Arfaoui, Y.; Brigaud, T.; Besbes, R. Synth. Commun. 2018, 48, 2242-2252

- 2017
-
1. Synthesis of C-Ribosyl-1,2,3-triazolyl Carboxamides. Solarte, C.; Dos Santos, M.; Gonzalez, S.; Miranda, L. S. M.; Guillot, R.; Ferry, A.; Gallier, F.; Uziel, J.; Lubin-Germain, N. Synthesis 2017, 49, 1993-2002.

2. Enantioselective Access to Robinson Annulation Products & to Michael Adducts Precursors: Where Do We Stand After 80 Years? Gallier, F.; Martel, A.; Dujardin, G. Angew. Chem. Int. Ed. 2017, 56, 12424–12458.
3.Tailored Approaches towards the Synthesis of L-S-(Trifluoromethyl)cysteine- and L-Trifluoromethionine-Containing Peptides. Gadais, C.; Saraiva-Rosa, N.; Chelain, E.; Pytkowicz, J.; Brigaud, T. Eur. J. Org. Chem. 2017, 246-251.
4. Structure-Activity Relationship Studies, SPR Affinity Characterization, and Conformational Analysis of Peptides That Mimic the HNK-1 Carbohydrate Epitope. Ieronymaki, M ; Nuti, F ; Brancaccio, D ; Rossi, G ; Real-Fernandez, F ; Cao, YH ; Monasson, O ; Larregola, M ; Peroni, E ; Uziel, J; Sabatino, G ; Novellino, E ; Carotenuto, A ; Papini, AM ; Rovero, P. ChemMedChem 2017, 12, 751-759.5. Synthesis of Ribavirin 2’-Me-C-nucleosides analogues. Cosson, F.; Faroux, A.; Baltaze, J.P.; Farjon, J.; Guillot, R.; Uziel, J.; Lubin-Germain, N. Beilstein J. Org. Chem. 2017, 13, 755-761.6. Structural Behavior of the Peptaibol Harzianin HK VI in a DMPC Bilayer: Insights from MD Simulations. Putzu, M.; Kara, S.; Afonin, S.; Grage, S. L.; Bordessa, A.; Chaume, G.; Brigaud, T.; Ulrich, A. S.; Kubar, T. Biophys. J. 2017, 112, 2602-2614.7. Homochiral versus Heterochiral Trifluoromethylated Pseudoproline Containing Dipeptides: A Powerful Tool to Switch the Prolyl-Amide Bond Conformation. Chaume, G.; Simon, J.; Lensen, N.; Pytkowicz, J.; Brigaud, T.; Miclet, E. J. Org. Chem. 2017, 82, 13602-13608.

- 2016
-
1. Incorporation of Trifluoromethylated Proline and Surrogates into Peptides: Application to the Synthesis of Fluorinated Analogues of the Neuroprotective Glycine-Proline-Glutamate (GPE) Tripeptide. Simon, J.; Pytkowicz, J.; Lensen, N.; Chaume, G.; Brigaud, T. J. Org. Chem. 2016, 81, 5381-5392.

2. Synthesis of protected enantiopure (R) and (S)-α-trifluoromethylalanine containing dipeptide building blocks ready to use for solid phase peptide synthesis. Devillers, E.; Pytkowicz, J.; Chelain, E.; Brigaud, T. Amino Acids 2016, 48, 1457-1468.3. Epitope mapping of anti-myelin oligodendrocyte glycoprotein (MOG) antibodies in a mouse model of multiple sclerosis: microwave-assisted synthesis of the peptide antigens and ELISA screening. Pacini, G.; Ieronymaki, M.; Nuti, F.; Sabatina, G.; Larregola, M.; Aharoni, R.; Papini, A. M.; Rovero, P. J. Pept. Sci. 2016, 22, 52-58.4. Functionalization of 2H-1,2,3-Triazole C-Nucleoside Template via N2 Selective Arylation. Lopes, A. B.; Wagner, P.; Alves de Souza, R. O. M.; Lubin Germain, N.; Uziel, J.; Bourguignon, J.-J.; Schmitt, M.; Miranda, L. S. M. J. Org. Chem. 2016, 81, 4540-4549.

5. Metal Promoted Diels-Alder Reactions. Gallier, F. Current Organic Chemistry, 2016, 20(21), 2222-2253.

6. The weight of flash chromatography: a tool to predict the mass intensity from thin-layer chromatography. Pessel, F.; Augé, J.; Billault, I.; Scherrmann, M-C. Beilstein J. Org. Chem. 2016, 12, 2351-2357.7. Access to Complex C2-Branched Glycoconjugates via Palladium-Catalyzed Aminocarbonylation Reaction of 2-Iodoglycals. Bordessa, A.; Ferry, A.; Lubin-Germain, N. J. Org. Chem. 2016, 81 (24), 12459–12465.